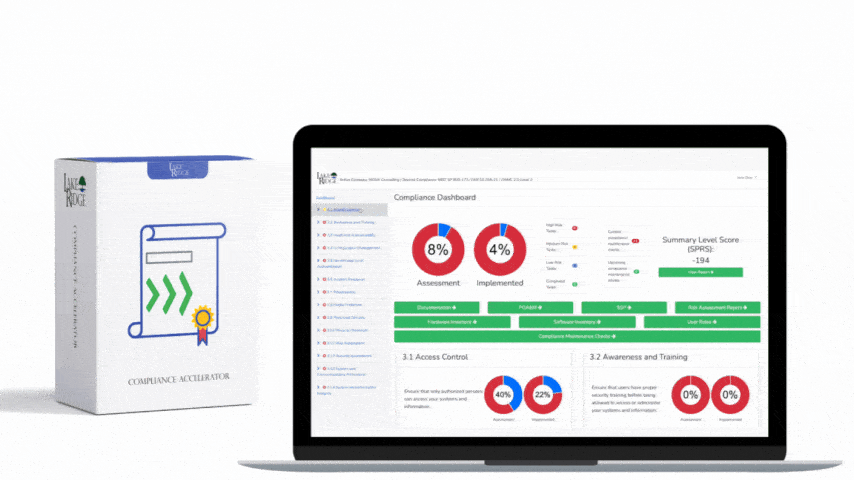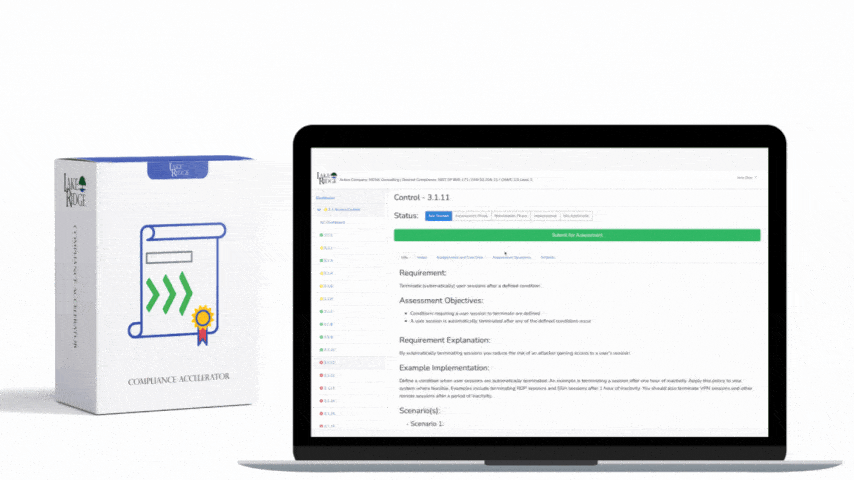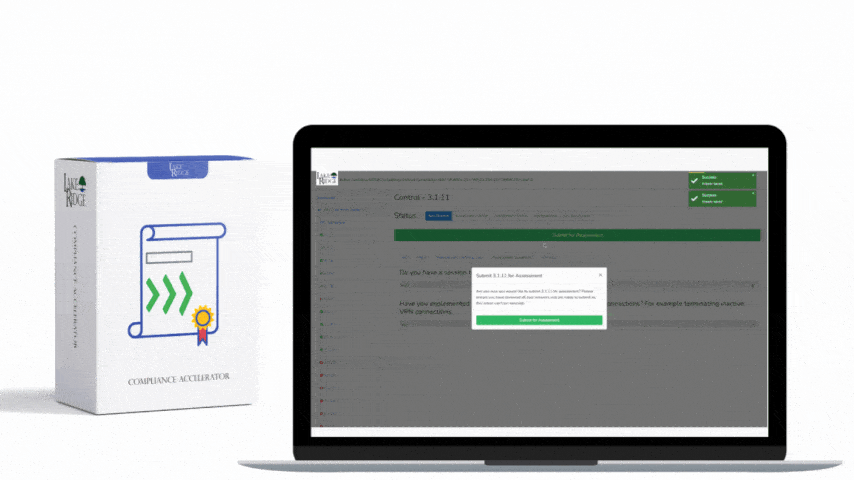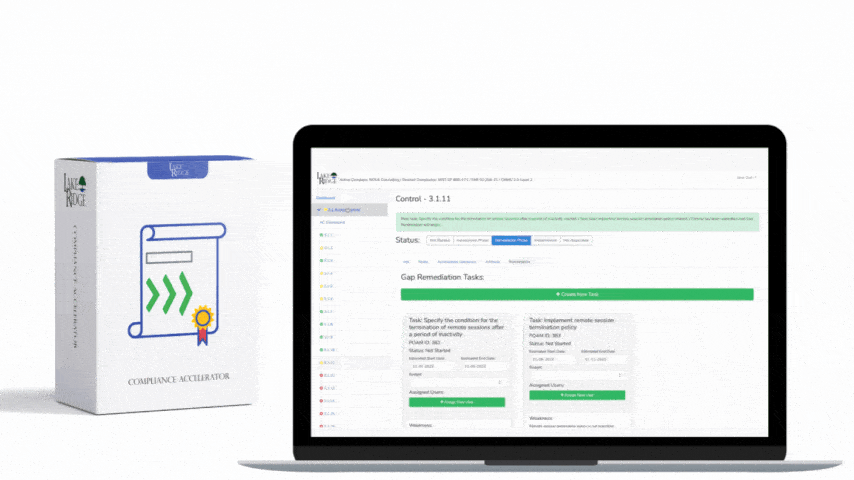
This post explains how small businesses can configure automated scanning tools and sanitization workflows to meet NIST SP 800-171 Rev.2 / CMMC 2.0 Level 2 control MA.L2-3.7.4 — sanitize or destroy diagnostic/test program media before reuse — with practical configuration steps, technical commands, and real-world examples.
Understanding MA.L2-3.7.4 and why automation matters
MA.L2-3.7.4 requires that diagnostic and test program media (for example, USB drives used for firmware debugging, SD cards in test fixtures, or internal storage in diagnostic appliances) be sanitized or destroyed before reuse so that Controlled Unclassified Information (CUI) cannot be recovered. The NIST SP 800-88 Rev.1 sanitization guidelines are the accepted technical reference for methods and validation. For a small business with limited staff, automated scanning and response reduces human error, provides repeatable evidence, and helps demonstrate compliance during audits.
How automated scanning tools fit into your compliance framework
Automated scanning tools do three critical things for this control: discover and inventory diagnostic/test program media, classify and tag media that may contain CUI or privileged diagnostic code, and trigger or perform an approved sanitization action (and log the result). Common tooling components include EDR/MDM agents, network and endpoint discovery, USB auditing utilities, asset management / CMDB, sanitization orchestration tooling (scripts, Ansible, SCCM/Intune runbooks), and centralized logging/SIEM for evidence retention.
Practical implementation steps (high level)
1) Asset Discovery & Inventory — Configure an automated discovery scan to identify removable and embedded storage across your environment. For Windows endpoints use MDM/EDR inventory (Intune, SCCM, CrowdStrike) and enable USB device enumeration; for Linux use osquery/Ansible + udev rules; for networked test equipment use SNMP or SSH-based inventory. Set discovery frequency to at least daily and immediately on device deprovision events.
2) Classification & Tagging — Create classification rules in the scanner: tag devices by VID/PID (USB), serial number patterns, mount points (/dev/sdX, /dev/nvme*), and hostname groups (lab-*, testbench-*). Maintain a list of approved diagnostic device types and mark unknown devices as high-risk. Use DLP or file-scanning modules to detect CUI file patterns (keywords, file extensions, metadata) on discovered media.
3) Automated Playbooks / Actions — Configure playbooks that respond to a flagged device: (a) quarantine/disable device access, (b) block further writes or network access, and (c) initiate a sanitization workflow. For Windows endpoints you can trigger a PowerShell runbook via Intune or SCCM; for Linux use an Ansible job that executes ATA / NVMe sanitize commands or calls a vendor sanitization API. Ensure tooling integrates with your ticketing system so every action has an approver and audit trail.
Specific technical configurations and commands
Choose sanitization methods per media type and document them in your Compliance Framework implementation notes. Examples of supported sanitization methods and commands:
- ATA Secure Erase (HDD/SSD): use hdparm:
hdparm --user-master u --security-set-pass P /dev/sdX hdparm --user-master u --security-erase P /dev/sdX- SCSI SANITIZE for enterprise drives: use sg_format/sg_sanitize tools; NVMe sanitize:
nvme format /dev/nvme0n1 -s1 nvme sanitize /dev/nvme0n1 --secure
- SSDs and eMMC: prefer vendor “crypto-erase” (secure-erase or NVMe sanitize) over overwrite due to wear-leveling; avoid DBAN style overwrites on SSDs. For USB sticks where vendor tools exist, use the vendor utility or a certified tool like Blancco; for lower-risk devices a full-disk overwrite with dd can be used:
dd if=/dev/zero of=/dev/sdX bs=1M status=progress(note: dd is slow and not fail-proof on SSDs).
- Encrypted volumes (BitLocker/FileVault): perform crypto-erase by deleting keys or using Manage-bde/Crypto APIs:
manage-bde -off C: Remove-BitLockerKeyProtector -MountPoint "C:" -KeyProtectorId <id>and then perform reformat if required. For Linux LUKS:
cryptsetup luksKillSlot /dev/sdX 0or re-key to render old data unrecoverable.
Real-world small business scenario
Example: a 30-person subcontractor has a test bench with SD cards and USB debugging sticks used by engineers. Implementation: deploy an MDM agent and osquery across endpoints to enumerate connected USB devices and capture VID/PID/serial. Create an automated rule: when a USB stick is marked “test-media” and flagged for reuse, trigger an Intune PowerShell runbook that copies a manifest, runs the vendor sanitization utility (or hdparm for SATA test boxes), verifies the return code, and sends the sanitized-report (device SN, method, timestamp, operator) to the SIEM and the IT ticket. For SD cards removed from a standalone bench PC, require chain-of-custody forms and sanitization at a controlled workstation running the approved script; keep the sanitized log attached to the ticket before reuse.
Verification, logging, and audit evidence
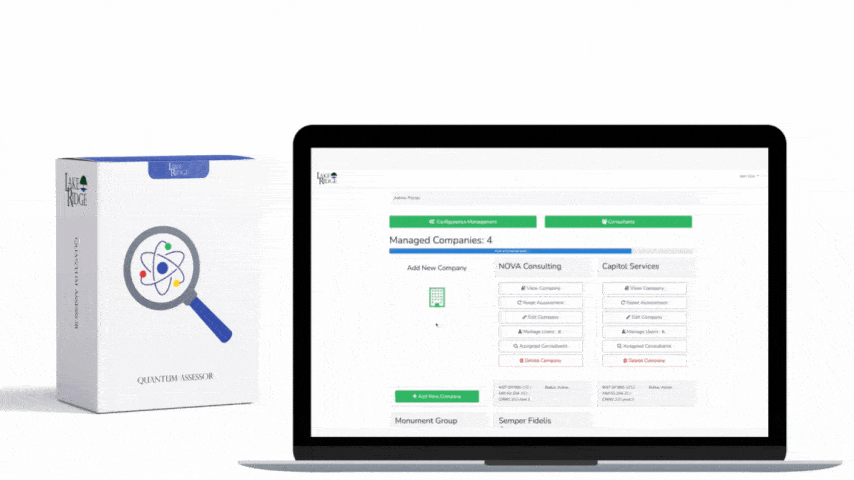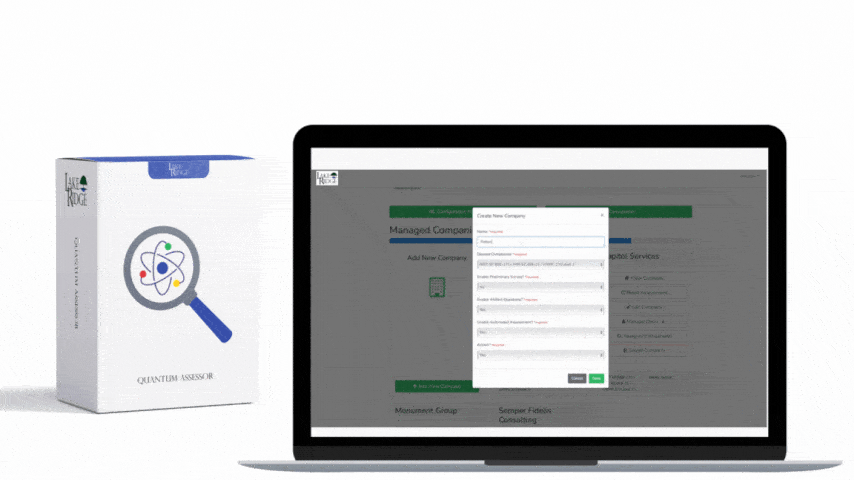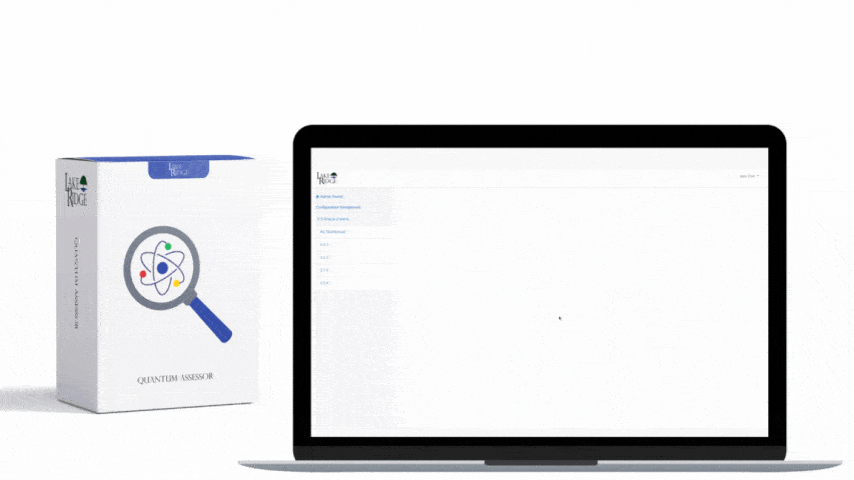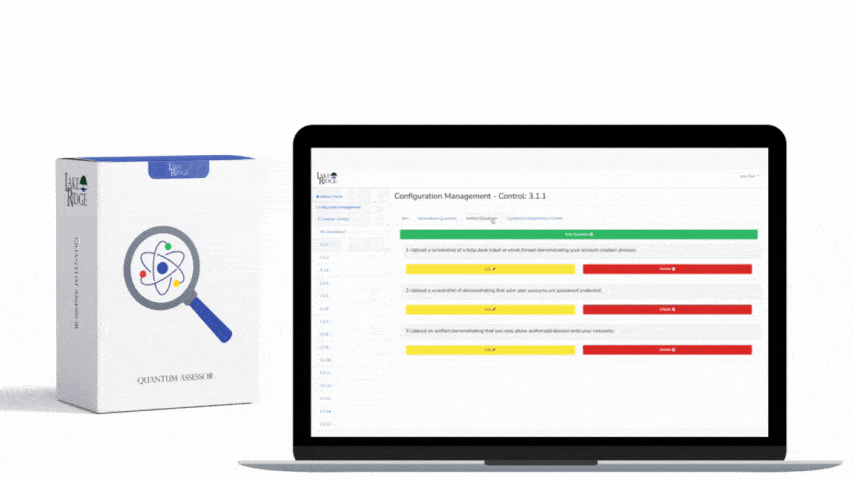
Automated tools must log who initiated the sanitization, the device serial, the method used, command return codes, and a hash or checksum of the device state where possible. Keep these logs in a tamper-evident store (forward them to SIEM or write-once storage). Periodically validate sanitization effectiveness with random forensic checks: mount a sanitized device in a forensics VM and run Autopsy or bulk_extractor to confirm no CUI remnants. Retain evidence per customer/contract requirements — common practice is to keep sanitization records for the lifecycle of the contract plus 3–6 years depending on DFARS clauses.
Compliance tips and best practices
- Map each media type to an approved sanitization method in your Compliance Framework documentation and make that mapping part of your automation playbooks. - Maintain an up-to-date asset/serial registry for all diagnostic/test devices so automation can match devices to required handling. - Use vendor-certified sanitization tools (Blancco, vendor utilities) for high-assurance needs and keep licensing and tool output as part of audit evidence. - Train staff and require manual approval for exceptions; automation should default to “quarantine until sanitized” for unknown devices. - Log everything: device identifiers, operator identity, method, verify result, retention location, and ticket ID.
Risk of not implementing: failing to sanitize diagnostic/test program media can lead to accidental disclosure of CUI (customer data, source code, debug builds), resulting in contract breach, regulatory penalties, reputational damage, and asymmetric risk where an attacker leverages leftover debug credentials to move laterally into sensitive systems. For SMEs working with DoD data, this could mean loss of contracts or serious compliance enforcement actions.
In summary, configuring automated scanning tools to satisfy MA.L2-3.7.4 means building a repeatable pipeline: discover and classify diagnostic/test media, automatically trigger an approved sanitization procedure tailored to media type, verify with forensic checks, and maintain immutable logs as evidence. For small businesses, combining MDM/EDR inventory, a CMDB, runbook automation (Intune/Ansible/SCCM), vendor sanitization tools, and SIEM logging will create an auditable, low-touch process that meets NIST/CMMC expectations while minimizing operational overhead.